More on Free Radicals
You got the three main components down: initiation, propagation and termination. These happen for all radical polymerizations, although new developments allow more control over their behavior. We'll get to fascinating topics like "living free radical polymerization" in the advanced course so come back for that later in detail or click here for a preview.
We're going to dive right in with the fascinating topics of back biting, short-chain branching, long-chain branching and what it means to be a hoop snake... Well, not really, since hoop snakes don't actually exist. The rest do, though, and they play a key role in determining the properties of LDPE. And in case you forgot how much polymer scientists love to come up with fun but sometimes cryptic short-hand notations for polymers, LDPE stands for "low density polyethylene." Which is not to be confused with "linear low density polyethylene" (LLDPE) (although many people do) but which is a totally different creature made a totally different way.
Radical Back Biting

"So tell me again what a hoop snake has to do with back biting?" Well, duh, it's just an analogy, a word picture to help you understand, or in this case, an actual picture. This mythical creature has the interesting ability of being able to grasp its tail in its mouth and then roll rapidly over hill and dale in search of prey or fleeing predators itself. How it actually propels itself has long been a mystery that we'll not attempt to solve here. The point of the analogy is this: back-biting.
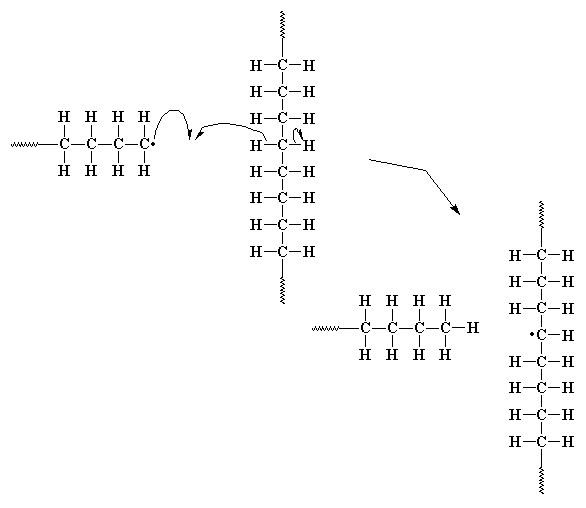
Remember that the propagating end of radical polyethylene is a primary radical as shown in the figure below on the left. Now this turns out to be the absolutely least stable free radical you can make with a hydrocarbon. It's so unstable that it will do just about anything to become more stable. Converting into a secondary (or even better, a tertiary) radical would help a lot. Under the conditions of this type of polymerization, the ethylene monomer is a solvent for the polymer being formed plus the temperature is way above the glass transition temperature of the final polymer. Both of these mean there's lots of molecular freedom, lots of molecular motion happening, and that means the oh-so-unstable chain end can come real close to the polymer chain that's already been formed. The figure below shows the whole glorious process.
Short-chain Branching
The interesting result of lots of motional freedom plus thermodynamics of hydrocarbon ring sizes is that multiple length branches can form. Because the chain end only bites back a few carbon atoms, the new secondary radical formed is maybe five to seven carbons from what was the chain end. Propagation starts up again from this secondary radical, and that leaves behind a short carbon chain dangling from the backbone. Got it?
And one thing that distinguishes LDPE from LLDPE is the length of these short-chain branches. In LDPE, they can be mostly five, six or seven carbons long, although some shorter and some longer do occur in small amounts. In LLDPE, the way it's made commercially, there are only even number branches. "Why?" you ask. Simple: LLDPE is made by copolymerizing ethylene with other alkenes like butene, hexene and octene. These give two-, four- and six-carbon branches. There's only one source I know of that provides odd-carbon alkenes (like pentene and heptene) and that would result in LLDPE with odd-carbon branches. Not much of this on the world market, that I'm aware of, so let's not worry about it here.
Long-chain Branching
Everyone, that is, except for the polymer chain which lost its hydrogen atom. It
now has not only one carbon atom with an unpaired electron, but two! Now
this looks bad but it's really not too difficult a problem, as it turns
out. The two carbon radicals, being right next to each other, can easily
join their unpaired electrons to form a pair, and thus form a chemical bond
between the two carbon atoms. Now the two atoms already shared one pair of
electrons, and the second shared pair creates a double bond at the end of the polymer chain.

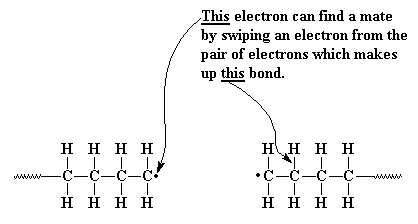
Sometimes, the unpaired electron at the end of a growing chain is so
unhappy that it will pair itself with an electron from a carbon-hydrogen bond
along the backbone of another polymer chain. This leaves an unpaired electron
which is nowhere near the propagating chain end. This electron can't form a
double bond the way the electron from the last example did, but it can and
will react with a monomer molecule, just the way the initiator fragment did.
This starts a new chain growing out of the middle of first chain! This is
called chain transfer to polymer, and the result is a branched polymer.
It is especially a problem with polyethylene, so much so that
linear non-branched polyethylene can't be made by free radical polymerization.
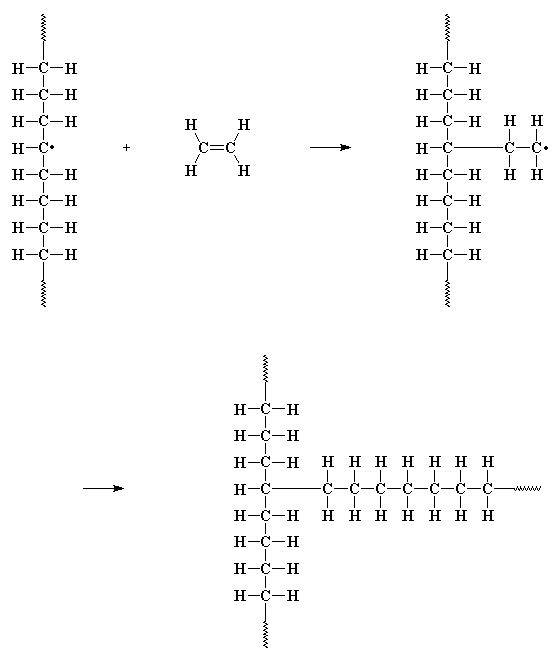
Such branching reactions have a big effect on how polyethylene behaves. And just so you can visualize all these branching options, the figure below shows both short- and long-chain branching for PE. Imagine trying to pack such chains close together so you get the maximum amount of inter-chain interaction. Not happening.

To find out how getting rid of branching helped make lowly polyethylene better than Kevlar® for use in bullet proof vests, visit the fun-filled, fact-filled and just plain fascinating polyethylene page.
The term "chain-growth" refers to how polymer chains are actually formed. With step-growth, all polymers grow slowly more or less at the same time and rate. With chain-growth, polymers are made one chain at a time. That is, whatever's in your reaction vessel is either monomer or high molecular weight polymer. There aren't any oligomers (unless you do things wrong like Neo did) and you could stop the reaction at any time, precipitate the polymeric component and filter it from the left over monomer, and it should be perfectly usable.
In actual fact, it's almost impossible to use up all the monomer in this type of polymerization. Why? Simple reason is that as conversion gets high, there's not enough monomers left to find the active chain end to add to it. The rate of polymerization grinds to a halt (so much for whetting your appetite) and you might as well just end it so you can get your polymer out.
Another very important aspect of this type of polymerization is that things happen fast. Chains literally "zip up" as fast as monomer can find an active chain end. Oh, but there can't be very many of those because they react with each other, at least for radical polymerizations.
Let me say that a different way: you only can start so many chains at any given time if you want high molecular weight. Start too many and the ends find each other, couple and they can't react any more. Problem is, the more active ends there are, the faster is conversion, which is desirable from an industrial perspective. But if the reaction goes too fast, things get out of hand: exotherms that vaporize monomer and blow the lid off your reaction vessel.
Confused yet? It's not really all that complicated. Why not make a list or a table with the characteristics and consequences of each type of polymerization. That will help you see key differences and why you do each the way you do so you get usable polymer without blowing anything up. I'd do it for you, but the exercise is good for you (heard that before, have you?). On to ROP.
Ring-opening- Some Combination of the Two
And of course, "ROP" is not some variant of "RIP" but rather "ring-opening polymerization." And here's where things aren't so clear cut. Most ROP's react with some initiator starting the process by opening one ring. The polymer grows only at the active chain end, it grows fast and at some point a side reaction or terminating molecule stops it... or not. Then another initiator starts another chain, and so on: one chain at a time just like for chain-growth. But here's the catch: the propagation step involves the type of intermediate and/or transition state found for step-growth polymerization plus the structure of the functional groups and polymer chain look like those of step-growth polymers.
And to make things even more complicated, it turns out you can usually make the polymer made via ROP by simple polycondensation using a step-growth mechanism. For example, take the caprolactone monomer in the example above, hydrolyze with water to give you an A-B monomer with an alcohol group at one end and carboxylic acid at the other. Now do the normal polyesterification reaction in which any alcohol can react with any acid to give a backbone ester and you have a step-growth polymer. More complicating is the fact that just looking at this polymer and the one made by chain-growth process wouldn't tell you which is which. Oh, sure, they would be clues in what kind of chain ends are present, but overall, the molecular compositions would be identical as would their properties. May seem confusing and like I said before, "it's complicated."
So which type of definition should I use, traditional or mechanistic? Depends on what day of the week it is, who you're talking to and what point you're discussing. One kind works fine for some general discussions while the other kind is more appropriate for others that are more detailed and specific. So just like ol' Neo, sink or swim, learn by doing, and practice trial by error; or put in your own trite phrase here. Time to move on to the next few sections where we look at individual families or examples of all three types of polymerizations. We'll delve into why a given polymerization mechanism for a given monomer or set of monomers gives the structures and properties that it does.
Right now, though, it would help you a lot to know what you DO know and more to the point, what you DON’T know. You should be starting to like taking quizzes, since they’re actually fun! And they really help you understand where you are in the learning process. So let’s take one, ok?
This quiz is about free radical polymerization. Don't be afraid to guess- your subconscious will often times pop up the right answer even if you're not sure of it: free radical quiz.
And when you're ready to move on, you can begin exploring the details of anionic vinyl polymerizations which are very different from their radical cousins. In fact, you could pick any of the other topics you'd like to pursue next; just go to the panel on the left at the top and click away...
